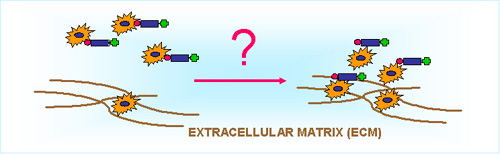
Adhesion is one of the most fundamental, iniciator steps of the cell-physiological cascade reactions which also involves chemotaxis and phagocytosis. Different families of extracellular matrix (elastine, collagene etc.) are synthesized by cells and expressed in association with characteristic partner molecules. Cell-ECM interactions have a major role in triggering chemotactic responses, cleavage of ECM molecules provides chemotactically active ligands. Derivatives of these molecules produced by enzymatic cleavage have a great significance in guiding healthy and malignant migratory cells.
In the focus of our "ECM-research" is to evaluate potential targeting potency of well characterized ECM sequences, like RGD, KTS and VGVAPG. This part of our experiments on chemotactic drug targeting (CDT) investigates the potency of different ECM sequences whether they have characteristic potnecy to modulate migratory effect of target cells (see below).
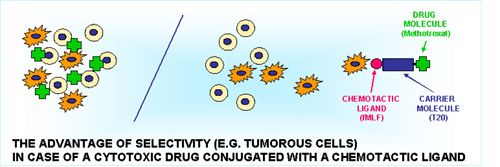
Characterization of the investigated sequences of the ECM and chemotaxis drug-targeting (CDT)
RGD
The RGD motif is found in a number of proteins that play a role in cell adhesion, including some forms of collagens, fibrinogen, vitronectin, von Willebrand factor, snake disintegrins and slime mold discoidins and serves as an adhesion ligand for members of the integrin family. Experimentally determined structures of cell-adhesion proteins reveal that the RGD motif is localized within loop regions and can adopt a broad set of conformations. The RGD sequence is also found in several important extracellular signal proteins as osteopontin or Aortic Preferentially Expressed Protein-1.
KTS
KTS-disintegrins are a subfamily of short monomeric disintegrins that are potent and selective inhibitors of alpha1beta1 integrin. Obtustatin, was purified from the venom of the Vipera lebetina obtusa viper. Obtustatin potently inhibits angiogenesis in vivo in chorioallantoic membrane of chicken and it reduces tumor development in Lewis lung syngeneic mouse model. Other disintegrins containing the KTS motif in their active integrin binding loop are viperistatin and lebestatin.
VGVAPG
A non-integrin receptor for ECM components is elastin binding protein (EBP), a specific binding site of VGVAPG motives of the elastin. The hexapeptide repeats in two-three segments in the C-terminal portion of the tropoelastin molecule. Chemotactic or MMP producing effect of elastin degradation products are transmitted by the 67kDa EBP galectin can be abolished by lactose addition.
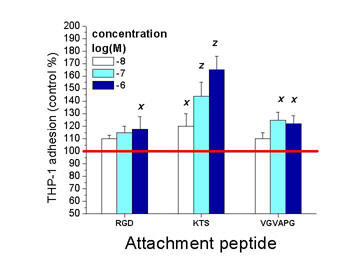
Our recent results demonstrate that tumorous monocytes have diverse, concenttration dependent affinity towards the three representative peptides of the ECM.
In adhesion assays the alpha1beta1 integrin specific KTS proved to be the most effective, while adhesion to surfaces coated with RGD sequences - possessing a similar, integrin specificity - was rather reduced. Inducer effect of the EBP specific VGVAPG suggest that responsiveness to different components of the ECM sequences is preseved in tumorous cells and may work as a coordinator factor in metstatic processes. (Kun, L. et al., FEBS, Budapest, 2005.)
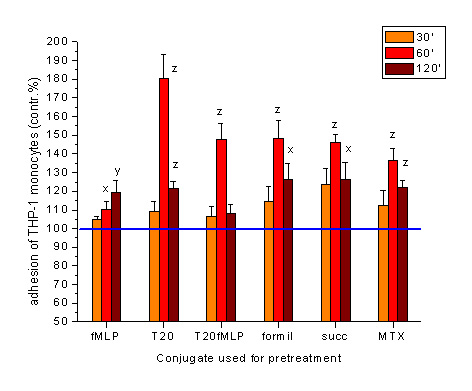
Investigations on the oligotuftsin (T20) based conjugates developed for chemotactic drug targeting (CDT) proved that however, itself the carrier can induce the highest adhesion of malignant monocytes, development of fMLP containing conjugate ((T20fMLP) or chemical midifications of the carrier with formyl or succinyl groups did not modify significantly the optimal effectiveness detected at 60 min.
Investigations of the methotrexate containing conjugate (MTX) indicate that the complete conjugate still possess the adhesion inducer effect which is essential for the successful targeting into of migrating/mobile subsets of metastatic cells. (Kun, L. et al. XXXV. MIT, Sopron, 2005.)